最近很多同学抱怨化学学不懂,好难应该怎么办😧,还有一个月就考试了。其实化学考的内容整体来说并没有像物理电磁那样特别考验逻辑,我认为其难的原因根本在于知识点太过琐碎(感觉可以跟隔壁统计坐一桌了)。所以,我有一个想法,我想要把内容整体给大家串一遍。既方便大家复习快速对应知识点出处,又能帮助还没有学的同学光速获取知识😂。所以,从这里,现在,开始了!!!
本文主要是对下面参考文献的翻译和整理,便于快速定位知识点和复习。有任何不妥、不清晰、不完善之处,还请查阅原文。
参考文献网址: https://apchem.org
第一单元
- 了解摩尔数$n$和相对原子质量$M$。
质谱仪
质谱仪画出的图测相对原子质量很爱考,下面是对这个图的具体描述:
- x-axis: Mass-to-charge ratio (m/z). For singly charged ions, this is effectively the mass of the isotope.
- y-axis: Relative abundance, showing how common each isotope is.
- Each peak represents an isotope of the element, and the height of the peak indicates its relative abundance.
大意就是有一个元素,通过质谱仪,由于一个元素有多种同位素,不同同位素有不同的相对原子质量,所以只要一个同位素被显现,就会产生一次波动。然后我们就可以进行计算。
The data from a mass spectrum allows calculation of the average atomic mass of an element by weighting the mass of each isotope by its relative abundance.
$$\text{Average Atomic Mass} = (\text{Mass}_1 \times \text{Abundance}_1) + (\text{Mass}_2 \times \text{Abundance}_2) + \text{...}$$
where abundances expressed as fractions, not percentages (其实就是加权平均数求期望值)。
我们通过硼(B)进行举例:

In this case, the two isotopes (with their relative abundances) are:
| Element | Relative Abundances |
|---|---|
| Boron-10 | 23 |
| Boron-11 | 100 |
The total mass of these would be $(23 \times 10) + (100 \times 11) = 1330$.
The average mass of these 123 atoms would be $1330 / 123 = 10.8$ (to 3 significant figures).
10.8 is the relative atomic mass of boron.
质谱图文献来源 https://www.chemguide.co.uk/analysis/masspec/elements.html
质谱图应用:(不太常考,可以先忽略)
- Isotope Identification: Confirms the presence and ratios of isotopes.
- Elemental Analysis: Determines the composition of unknown samples.
- Molecular Studies: Mass spectrometry can be extended to molecules, revealing their structure and composition.
- Forensics and Medicine: Used in drug testing, carbon dating, and medical diagnostics.
纯净物元素组成
Pure substance is classified as elements (单质) and compound (化合物). 因此我们可以通过此推出相对分子质量和元素在相对分子质量中的占比:
公式:
$$\text{Percent by Mass of Element} = \frac{\text{Mass of Element in 1 Mole of Compound }}{ \text{ Molar Mass of Compound}} × 100$$
依旧加权平均数。以水举例:
Water ($\ce{H2O}$)
- Molar mass of $\ce{H2O} = \ce{(2 \times 1.01 g/mol) + (16.00 g/mol) = 18.02 g/mol}$
- Mass of $\ce{H = 2.02 g/mol}$
- Mass of $\ce{O = 16.00 g/mol}$
- Percent by mass of $\ce{ H = (2.02 / 18.02) × 100 ≈ 0.112 }$
- Percent by mass of $\ce{ O = (16.00 / 18.02) × 100 ≈ 0.888 }$
还有两个分子式,但不太重要,就不细说了:
- Empirical formula: The simplest whole-number ratio of atoms in a compound.
- Molecular formula: The actual number of atoms of each element in one molecule.
For example, glucose has a molecular formula of $\ce{C6H12O6}$ but an empirical formula of $\ce{CH2O}$.
大意就是葡萄糖可以被两种式子所写。
混合物元素组成
混合物分为均匀混合物和非均匀混合物:
- Homogeneous mixtures: These have a uniform composition throughout. They are often called solutions. Examples include salt water, air, and alloys like brass.
- Heterogeneous mixtures: These do not have a uniform composition. Different parts of the mixture can be seen and separated physically. Examples include oil and water, sand in water, or salad.
Mass/volume percent 和 mole fraction 同为加权平均数。
Molarity ($M$) is defined as the number of moles of solute per liter of solution.
这个还挺重要的,因为之后溶液的相对摩尔浓度都用这个$M$作为单位。
Molality ($m$) is the number of moles of solute per kilogram of solvent.
公式:
$$\text{m} = \frac{ \text{Moles of Solute} }{ \text{Kilograms of Solvent} }$$
Molality is independent of temperature, making it useful in studies involving colligative properties.
原子组成
- 质子(proton)positively charged 带正电
- 中子(neutron) neutral 不带电
- 电子(electron) negatively charged 带负电
- 原子核(nucleus)= 质子 + 中子
原子排布
原子核外排布着一圈又一圈的电子轨道,不同的轨道蕴含着不同的能量和不同互谅的电子,电子轨道的volume从里向外逐渐增大。从里向外依次是s、p、d、f。
The main types of orbitals are s, p, d, and f:
- s orbitals are spherical and can hold up to 2 electrons.
- p orbitals are dumbbell-shaped and can hold up to 6 electrons.
- d orbitals can hold up to 10 electrons.
- f orbitals can hold up to 14 electrons.
电子在轨道上的排布规律必须遵循3个定律:
- Aufbau Principle: Electrons fill the lowest-energy orbitals first before moving to higher-energy orbitals. 先填满里面的,再填外面的
- Pauli Exclusion Principle: Each orbital can hold a maximum of two electrons, and these must have opposite spins. 每个小轨道上的电子排布⬆️⬇️
- Hund’s Rule: When filling orbitals of the same energy, electrons occupy them singly with parallel spins before pairing up.
These rules ensure that electrons are distributed in the most stable arrangement possible.
其实就相当于s有一个小房间,p有3个,d有5个,f有7个,每个小房间最多只能有两个电子。
书写电子构型 Writing Electron Configurations
Electron configurations are written using numbers and letters that indicate the energy level and type of orbital, along with superscripts to show the number of electrons.
在第几族就表示s或者p或者d同族的前面的系数(比如2s前面的2),轨道上面的数字标表示在这个小轨道上的有几个电子(比如3d⁶表示在3d这个小轨道上有6个电子排布)
- Hydrogen (Z = 1): 1s¹
- Oxygen (Z = 8): 1s² 2s² 2p⁴
- Iron (Z = 26): [Ar] 4s² 3d⁶(这里Ar开头意味着前面的电子排布轨道和Ar相同,后面跟着的是在此基础上叠加电子所形成的产物)
来看看此玩意在元素周期表中的运用
- Elements in the same group have similar outer electron configurations, leading to similar chemical properties. 同族元素有相似外电子排布例如氢和锂外部都是s¹,所有化学性质相似
- Moving across a period adds electrons to the same energy level, gradually filling orbitals. 同周期元素从左向右依次在亚轨道和小房间中累加电子
- Moving down a group adds new shells, increasing the size of atoms and changing their reactivity. 从一族移到下一族增加一个原子层,增加原子半径
Photoelectron Spectroscopy 光电子光谱
- Ionization Process: In PES, a photon (usually from ultraviolet or X-ray radiation) hits an atom or molecule, causing an electron to be ejected.
- Kinetic Energy Measurement: The energy of the ejected electron is measured. The binding energy of the electron is calculated using the equation:
Binding Energy = Photon Energy − Kinetic Energy of Electron
- Information Obtained: The binding energy reflects the energy required to remove an electron from a particular orbital, providing insight into the electronic configuration of the atom or molecule.
这玩意也挺容易考的
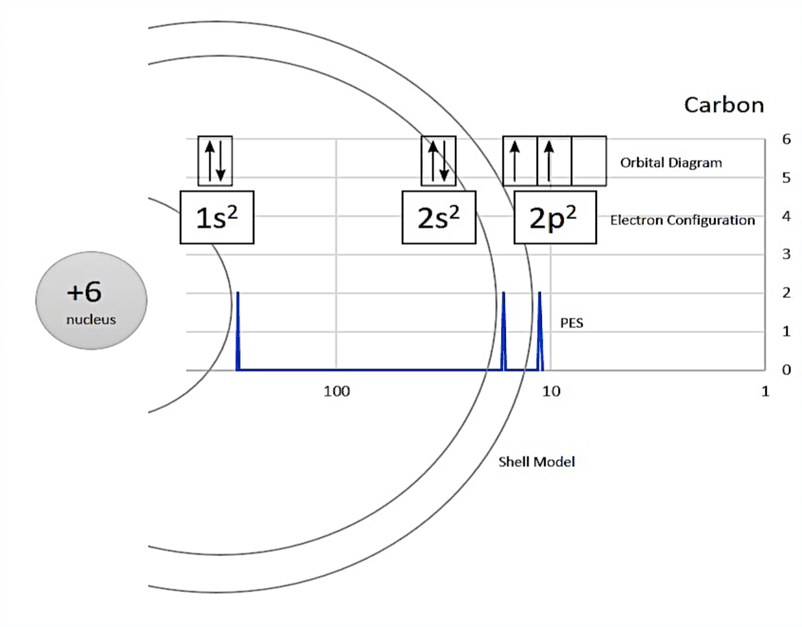
主要应用在于根据亚电子轨道来判断此是何物。
这个图,从左往右依次是1s, 2s, 2p...
然后注意观察纵坐标吧,作者找的这个图峰高都是一样的,假如不一样,应当通过矮的和高的对比来看出亚轨道上面的数字标究竟是多少。
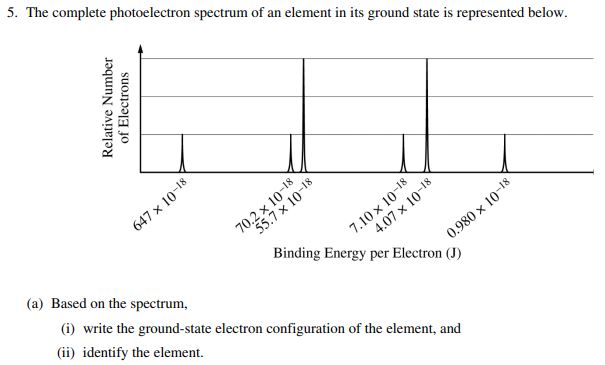
这个更复杂,但其实就是考试原题
答案:1s² 2s² 2p⁶ 3s² 3p⁶ 4s²
这个直到最后一个是4s²,所以这个物质是$\ce{Ca}$。
题目和图片来源 https://fiveable.me/ap-chem/unit-1/photoelectron-spectroscopy/study-guide/Xx7nwanr96Uzt1zSvwRA
元素周期表的一些基本规律
Atomic Radius 原子半径
- Trend down a group: Atomic radius increases because new electron shells are added, increasing the distance between the nucleus and the outermost electrons.
- Trend across a period: Atomic radius decreases from left to right. Although more protons are added to the nucleus, pulling electrons inward, no new shells are added, so the effective nuclear charge increases and the atom becomes smaller.
- 同族从上到下半径从小到大
- 同周期从左向右半径从大到小
Ionization Energy 电离能
Ionization energy is the energy required to remove an electron from an atom.
- Trend down a group: Ionization energy decreases because the outer electrons are farther from the nucleus and experience less attraction.
- Trend across a period: Ionization energy increases from left to right due to stronger nuclear attraction as effective nuclear charge increases.
- 同族从上到下电离能减小
- 同周期从左往右电离能增大
Electronegativity 电负性
Electronegativity is the ability of an atom to attract shared electrons in a bond.
- Trend down a group: Electronegativity decreases as atomic size increases, reducing the pull on bonding electrons.
- Trend across a period: Electronegativity increases from left to right as atoms become smaller and nuclear charge increases.
- 族从上到下电负性减小
- 周期从左往右电负性减小
Electron Affinity 电子亲合能
Electron affinity measures the energy change when an atom gains an electron.
- Trend down a group: Electron affinity generally becomes less negative (less favorable) due to increased distance between the nucleus and the added electron.
- Trend across a period: Electron affinity generally becomes more negative (more favorable), especially for halogens, as atoms more strongly attract extra electrons to complete their valence shell.
电子亲合能…
- …同族从上到下更弱
- …同周期从左往右更强
Metallic Character 金属性
Metallic character refers to the tendency of an element to lose electrons and form cations.
- Trend down a group: Metallic character increases because ionization energy decreases.
- Trend across a period: Metallic character decreases from left to right as elements become less likely to lose electrons and more likely to gain them.
金属性…
- …从上到下增加
- …从左往右减少
一个元素的价电子(Valence Electrons)实际上就是最外层电子数。
举例:
- Group 1 (alkali metals) have 1 valence electron and readily form +1 cations.
- Group 17 (halogens) have 7 valence electrons and tend to gain 1 electron to form -1 anions.
为了形成化合价,必须达到8电子稳定结构。有的元素得电子很容易,有的元素失电子很容易,具体情况具体分析。原子得失电子后就变成了正负离子,正负离子相结合就变成了ionic compounds
离子的形成
- Metal loses electrons → forms a cation
- Nonmetal gains electrons → forms an anion
- Electrostatic attraction between cation and anion forms an ionic bond
离子化合物性质 Properties of Ionic Compounds
- High melting and boiling points due to strong ionic bonds
- Solid at room temperature with crystalline structures
- Conduct electricity when molten or dissolved in water, because ions are free to move
第一单元终于结束了,等作者歇会儿就发第二个……
(((o(゚▽゚)o)))