OK,前面的单元全是对化学物质的定义和解释,从这一单元开始,才真正开始AP化学的学习。😁
第四单元
Introduction of reactions
- 化学反应是反应物转化为化学组成不同的产物的过程。
- 理解化学反应是化学的基础,可解释物质间相互作用、结合与变化。
Key Concepts
- Reactants: Starting substances in a chemical reaction
- Products: Substances formed after the reaction
- Chemical Equation: Symbolic representation of a reaction
- Example: $\ce{2H2 + O2 -> 2H2O}$
总而言之,箭头左边的是反应物,箭头右边的是生成物。
Types of Reactions
Synthesis (Combination): 多合一
- Two or more substances combine to form a single product
- $\ce{A + B -> AB}$
Decomposition: 一分多
- A single compound breaks down into simpler substances
- $\ce{AB -> A + B}$
Single Replacement (Displacement): 单置换反应。反应物和生成物都必须要是单质+化合物。
- An element replaces another in a compound
- $\ce{A + BC -> AC + B}$
Double Replacement (Metathesis): 复分解反应。两个化合物双双互换粒子。
- Exchange of ions between two compounds
- $\ce{AB + CD -> AD + CB}$
Combustion: 燃烧反应
- A substance reacts with oxygen, producing energy
- $\ce{ \text{Hydrocarbon} + O2 -> CO2 + H2O}$
Law of Conservation of Mass
- Matter is neither created nor destroyed in a chemical reaction
- Total mass of reactants = total mass of products
- 反应物质量 = 生成物质量
Net Ionic Equations
- 净离子方程式只显示实际参与化学反应的粒子。
- 省略旁观离子(反应前后不变的离子)。
Key Concepts
- Spectator ions (没有参与实际反应的离子): Ions present in solution that do not take part in the reaction
- Molecular equation (展示所有粒子): Shows all reactants and products as compounds
- Ionic equation (展示所有可以分开的强电解质离子): Shows all strong electrolytes dissociated into ions
- Net ionic equation (只展示参与反应的离子): Eliminates spectator ions to focus on the chemical change
Steps to write net ionic equations
- 写出配平的分子方程式
- 将所有强电解质拆分为离子形式
- 找出并消去旁观离子
- 写出最终的净离子方程式
Example
- Molecular equation:
$$\ce{NaCl(aq) + AgNO3(aq) -> AgCl(s) + NaNO3(aq)}$$ - Ionic equation:
$$\ce{Na+(aq) + Cl-(aq) + Ag+(aq) + NO3-(aq) -> AgCl(s) + Na+(aq) + NO3-(aq)}$$ - Net ionic equation:
$$\ce{Ag+(aq) + Cl-(aq) -> AgCl(s)}$$
Representations of Reactions
其实这一部分没什么要讲的,就是让你知道配平的分子式和离子式还有方程式都能表示化学反应。
Physical and Chemical Changes
在这一部分我们主要是定义一下物理变化和化学变化的实质。
Physical Changes
指物质的状态发生变化,一般都是可逆的。
- Change in state or appearance without altering chemical composition
- Reversible in many cases
- Examples: Melting, boiling, freezing, dissolving, breaking, cutting
Chemical Changes
指产生了新物质具有不同化学性质,变化不可逆。
- Change that produces a new substance with different chemical properties
- Often irreversible under normal conditions
- Indicators: Color change, gas formation, precipitate formation, energy release/absorption
- Examples: Rusting of iron, combustion, baking a cake, digestion
Distinguishing Between Changes
| Feature | Physical Change | Chemical Change |
|---|---|---|
| Compostion | Unchanged | Altered |
| Reversibility | Often reversible | Usually irreversible |
| Energy Change | Minor | Often Significant |
| Example | Ice melting, sugar dissolving | Iron rusting, wood burning |
Stoichiometry
我们可以基于已经平衡了的化学式,来计算出每个反应物和生成物的物质的量($n$)。
Key concepts
- Mole Ratios: Derived from the coefficients in a balanced equation
- Limiting Reactant: Reactant that determines the maximum amount of product
- Excess Reactant: Reactant left over after the reaction is complete
- Theoretical Yield: Maximum amount of product predicted by stoichiometry
- Actual Yield: Amount of product actually obtained
- Percent Yield: $\frac{\text{Actual Yield} }{ \text{Theoretical Yield} } \times 100\%$
Steps to Solve Stoichiometry Problems
- 书写并配平化学方程式
- 将已知量换算为物质的量(摩尔)
- 利用摩尔比计算目标物质的量
- 把摩尔数换算回克、升或分子数等单位
- 核对计算过程与单位一致性
Example
Given:
$$\ce{2H2 + O2 -> 2H2O}$$
- If 4 moles of $\ce{H2}$ react with 2 moles of $\ce{O2}$:
- $\ce{H2}$: $$\ce{4 \text{moles} \times \frac{ 2 \text{moles} H2O }{ 2 \text{moles} H2 } = 4 \text{moles} H2O}$$
- $\ce{O2}$: $$\ce{2 \text{moles} \times \frac{ 2 \text{moles} H2O }{ 1 \text{mole} O2 } = 4 \text{moles} H2O}$$
- Both reactants are perfectly stoichiometric; 4 moles of water form.
Introduction to Titration
这个方法酸碱中和反应中特别爱考。
滴定是用已知浓度溶液与待测溶液反应测定待测溶液的浓度。
但这一部分主要介绍滴定实验如何做,和在什么反应下用滴定可以很好的解决问题。具体可能会在之后的酸碱单元进行深究。
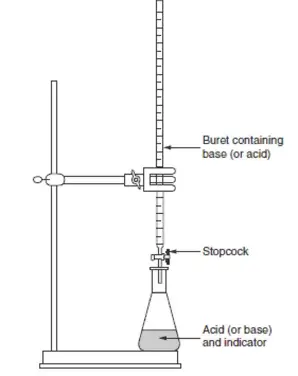
Key Concepts
- Analyte: 浓度未知。
The solution whose concentration is unknown. - Titrant: 浓度已知,需要其与待测物反应。
The solution of known concentration used to react with the analyte. - Equivalence Point: 滴定剂恰好完全中和待测物。
Point where the amount of titrant exactly neutralizes the analyte. - End Point: 指示剂变色指示。
Point indicated by a color change of an indicator, close to the equivalence point.
Types of Titrations
不咋考。
Acid-Base Titration
- Determines concentration of an acid or base
- Common indicators: phenolphthalein, bromothymol blue
Redox Titration
- Involves electron transfer reactions
- Example: Titration of $\ce{Fe^{2+}}$ with $\ce{KMnO4}$
Complexometric Titration
- Forms complexes with metal ions
- Example: EDTA titrations for hardness of water
Precipitation Titration
- Formation of a precipitate signals completion
- Example: $\ce{AgNO3}$ titration for $\ce{Cl-}$ ions
Types of Chemical Reactions
这一部分又开始放水了,主要是和前面讲的化学反应的类型基本一样,就只是再多引入了一个氧化还原反应而已。
Redox Reactions
- Involves transfer of electrons (oxidation-reduction)
- Example: $$\ce{2Na + Cl2 -> 2NaCl}$$
Introduction to Acid-Base Reactions
这一部分也只是主要提前预告一下酸碱反应,第八单元才会重点学习。
Acid-base reactions involve the transfer of protons ($\ce{H+}$ ions) between reactants. These reactions are central in chemistry, biology, and environmental processes.
酸碱反应的本质是反应物之间 $\ce{H+}$ 的转移。
Key concepts
- 酸:作为反应物可以电离出氢离子和共轭碱(供给氢离子)
- 碱:在反应物中接受氢离子并生成共轭酸(消耗氢离子)
- pH:衡量氢离子浓度,计算公式为$\ce{pH = -log[H+]}$
- pOH:衡量氢氧根离子浓度,计算公式为$\ce{pOH = -log[OH-]}$
Types of Acid-Base Reactions
Strong Acid + Strong Base: Complete dissociation in water
- Example: $$\ce{HCl + NaOH -> NaCl + H2O}$$
Weak Acid + Strong Base: Partial dissociation of weak acid
- Example: $$\ce{CH3COOH + NaOH -> CH3COONa + H2O}$$
Strong Acid + Weak Base:
- Example: $$\ce{HCl + NH3 -> NH4Cl}$$
- Neutralization Reactions: Formation of water and a salt from an acid and a base
Indicators
常见的酸碱指示剂。
- Substances that change color depending on pH
- Examples: Litmus paper, phenolphthalein, bromothymol blue
Oxidation-Reduction (Redox) Reactions
分子中有原子的化合价在经历了某种反应后会升高/降低,我们把这类反应叫做氧化还原反应。
Key Concepts
[!NOTE]
发生氧化反应时,反应物为还原剂;发生还原反应时,反应物为氧化剂
- Oxidation: Loss of electrons (increase in oxidation state)
- Reduction: Gain of electrons (decrease in oxidation state)
- Oxidizing agent: Species that is reduced, causes oxidation
- Reducing agent: Species that is oxidized, causes reduction
Identifying Redox Reactions
- Assign oxidation numbers to all elements in the reaction.
- Look for changes in oxidation numbers.
- Determine which species is oxidized and which is reduced.
[!NOTE]
如何判断其反应类型和某原子得失状况的口诀
升失氧,降得还。
化合价升高,还原剂失去电子,发生氧化反应;后一句反之亦然。
Example
可以用上面那个口诀进行试验。
$$\ce{Zn(s) + Cu^{2+}(aq) -> Zn^{2+}(aq) + Cu(s)}$$
- $\ce{Zn}$: $\ce{0 -> +2}$ (oxidation)
- $\ce{Cu}$: $\ce{+2 -> 0}$ (reduction)
- $\ce{Zn}$ is the reducing agent; $\ce{Cu^{2+}}$ is the oxidizing agent
Types of Redox Reactions
在一般情况下,我们认为只要化学反应后出现了化合价升高/降低,无论什么其他的反应类型。都叫其氧化还原反应。
- Combination reactions: Two elements combine, electrons are transferred
- Decomposition reactions: Single compound breaks down, electron transfer occurs
- Displacement reactions: One element replaces another in a compound
- Combustion reactions: Rapid oxidation producing energy